https://dx.doi.org/10.24016/2026.v12.449
ORIGINAL ARTICLE
Association between quality of life with depressive symptoms, anxiety,
stress and emotional distress in Peruvian cancer patients: a cross-sectional
study
Sofía C. Malaquias-Obregon 2, David Villarreal-Zegarra 2,
Anthony Copez-Lonzoy 1*, Jackeline
García-Serna 2, Milagros Cabrera-Alva 2, Ana L.
Vilela-Estrada 2
1 Universidad
San Ignacio de Loyola, Unidad de Investigación en Bibliometría, Peru
2 Instituto
Peruano de Orientación Psicológica, Lima, Peru.
* Correspondence: anthonycopez22@gmail.com
Received: February 23, 2025 | Revised: August 16, 2025 | Accepted: March 02, 2026 | Published Online: March 26, 2026.
CITE IT AS:
Malaquias-Obregon, S. C., Villarreal-Zegarra, D.,
Copez-Lonzoy, A., García-Serna, J., Cabrera-Alva, M., & Vilela-Estrada, A.
L. (2026). Association
between quality of life with depressive symptoms, anxiety, stress and emotional
distress in Peruvian cancer patients: A cross-sectional study. Interacciones, 12, 449. https://doi.org/10.24016/2026.v12.449
ABSTRACT
Introduction: Cancer patients frequently experience psychological
symptoms such as depression, anxiety, perceived stress, and emotional distress,
which may negatively affect their quality of life. However, the impact of
comorbid mental health symptoms on both mental and physical components of
quality of life remains insufficiently explored in Latin American populations.
Objective: To determine the association between depressive
symptoms, anxiety, stress, and emotional distress (exposures) and quality of
life (outcome) in Peruvian cancer patients.
Method: A cross-sectional study was conducted among 465 adult
cancer patients recruited from oncology services. Mental health variables were
dichotomized according to established cut-off points. Quality of life (mental
and physical components) was categorized into low–medium versus high levels.
Generalized linear models with Poisson distribution and robust variance were
used to estimate crude and adjusted prevalence ratios (PR) with 95% confidence
intervals.
Results: A prevalence of anxious symptoms of 27.5%, depressive
symptoms of 20.4%, symptoms of moderate-severe stress of 83.2%, and emotional
discomfort of 57.4% were identified. Regarding the Poisson regression model,
cancer patients with 12 or more years of education were less likely to have
symptoms of emotional distress. Moderate to severe depressive symptoms,
anxiety, stress, and emotional distress were significantly associated with
low–medium mental quality of life. Comorbidity of psychological symptoms showed
a stronger association with impaired mental quality of life compared to
individual symptoms.
Conclusions: Emotional distress and stress were found to be the
main variables associated with both physical and mental quality of life. Other
variables associated with mental health problems include a high level of
education. Finally, it was found that lower quality of life (both mental and
physical) is associated with greater comorbidity of mental health problems.
Keywords: Mental health, Quality of life, Cancer, Physiological effects.
INTRODUCTION
Cancer is one of the leading causes of morbidity and
mortality worldwide (IHME & Global Burden of Disease, 2024; IHME Global
Burden of Disease, 2024). By 2020, there were more than 19 million new cases
and nearly 10 million deaths for cancer, and projections indicate a substantial
increase in both incidence and mortality by 2040 (Cancer (IARC) & The
International Agency for Research on Cancer, 2022; Instituto Nacional del Cáncer, 2015). Approximately 70% of cancer deaths occur in
low- and middle-income countries; in these contexts, this situation is
particularly worrying due to population aging and the persistence of
preventable risk factors (Cancer (IARC) & The International Agency for
Research on Cancer, 2022; Organización Mundial de la salud,
2021).
Beyond its epidemiological impact, cancer represents a
significant burden on patients' quality of life, particularly in low- and
middle-income countries, where access to comprehensive cancer care remains
limited (Cardone & Arnold, 2023; Ngo et al., 2023). Given that physical and
mental health are essential components of quality of life, they play a
fundamental role in overall well-being and in people's ability to lead full and
satisfying lives (Jamil et al., 2023). In this regard, cancer patients in low-
and middle-income countries often experience a decline in their quality of life
because of symptom burden, adverse treatment effects, financial toxicity, and
inadequate support systems(Adekunle et al., 2025;
Ahmad et al., 2025).
In Peru, cancer poses a major public health challenge
within a fragmented healthcare system centered in Lima, where many patients
face delays in diagnosis, limited access to specialized treatments, and
insufficient psychosocial support (Hernández-Vásquez et al., 2025). These
conditions can exacerbate the physical and emotional burden of the disease,
increasing vulnerability to poor quality of life among cancer patients (Bergerot et al., 2024; Bustamante et al., 2022; Vy et al.,
2025).
The experience of receiving a cancer diagnosis (Cao et
al., 2017; Fernández de Larrea-Baz et al., 2020) and undergoing treatment
(American Cancer Society, 2022; Trayes & Cokenakes,
2021), often involves profound physical and psychological stress for patients
and their families due to its association with suffering and death. In this
regard, the stage and type of treatment, along with other factors such as the
type of cancer, age, sex, and social and environmental conditions (rural
residence, illiteracy, and low income), influence the deterioration of
patients' physical and psychological functioning, affecting various areas such
as family, work, social activities, and emotional and sexual well-being, among
others (Wen et al., 2019). Common physical symptoms include fatigue, pain, and
insomnia (Berger et al., 2010; Chiu et al., 2015; Fuller et al., 2018; Nipp et
al., 2017), while psychological symptoms such as depression, anxiety, and
stress are highly prevalent (Antoni & Dhabhar,
2019; Linden et al., 2012; Niedzwiedz et al., 2019; Weber & O’Brien, 2017),
particularly among hospitalized patients and those facing substantial
treatment-related financial burdens (Gilligan et al., 2018; Lu et al., 2019;
Niedzwiedz et al., 2019).
Scientific evidence indicates that the physical,
psychological and social problems faced by people with cancer can interfere
with their recovery process and significantly deteriorate their quality of life
(Weber & O’Brien, 2017). Several studies have demonstrated that this is
influenced by individual, social and clinical factors, such as the type and
stage of the illness and the treatment received. In particular, the presence of
psychological symptoms such as anxiety, depression and stress, unsatisfied needs
and advanced stages of cancer are consistently associated with a lower quality
of life (Hu et al., 2021; Li et al., 2018; Mace et al., 2021; Ngan et al.,
2021). In this vein, patients with breast cancer who present a greater burden
of psychological distress and uncertainty have lower quality of life scores, on
the contrary, those who show greater awareness of the illness and active
emotional strategies (Ngo et al., 2023).
People with cancer may have co-morbidities, which are
additional conditions or disorders that coexist with the primary disease
(Feinstein, 1970; Sarfati et al., 2016). Such comorbidities affect the
development and treatment of people with cancer (Sarfati et al., 2016). Also,
cancer patients may have more than one co-morbidity, including a combination of
depression and anxiety (Jeffery et al., 2019; Kugbey
et al., 2020). This comorbidity of mental health problems allows us to consider
transdiagnostic intervention as a viable alternative in the care of nuclear
vulnerabilities in cancer patients (Mirapeix, 2017; Tortella Feliu, 2014). On the other hand, the high level of
education, high monthly income, adequate care, interventions and early
treatment favor the quality of life of cancer patients (Dunne et al., 2017;
Ngan et al., 2021). Therefore, it must be ensured that patients receive
psychoeducation and palliative care to reduce physical and psychological
symptoms (Matsuda et al., 2014; Zhuang et al., 2018). Based on the exposed
evidence, the sociodemographic factors are considered for the statistical
analysis of this study.
Most studies have concentrated on specific
psychological conditions and have been carried out in high-income nations,
despite mounting evidence from around the world showing a connection between
mental health, including depressive symptoms, anxiety, stress, and emotional
distress, and quality of life in cancer patients. Only a small number of
studies have systematically measured psychological distress, with reported
prevalences ranging from 20% to 70%, despite the rising cancer burden in low-
and middle-income countries (where roughly 50% of the world's population
resides), this is partly due to a lack of funding and culturally appropriate
assessment tools (Bergerot et al., 2024; Walker et
al., 2021). Additionally, while research from high-income nations confirms that
cancer patients experience high rates of anxiety and depression, the evidence
from low- and middle-income nations is still limited and inconsistent, which
hinders the development and application of contextualized and successful
psychological interventions (Nakie et al., 2024).
To address this gap, we conducted a cross-sectional
study to determine the association between depressive symptoms, anxiety,
stress, and emotional distress (exposures) and quality of life (outcome) in
Peruvian cancer patients. Secondary objectives include determining the
association between educational level, type of cancer, and clinical stage and
mental health problems in Peruvian cancer patients, as well as analyzing the
comorbidity of mental health problems and their association with quality of
life in Peruvian cancer patients. In line with the objective, the hypothesis
was proposed that depressive symptoms, anxiety, stress, and emotional distress
have a significant association with quality of life in Peruvian cancer
patients.
METHODS
Study design
This cross-sectional study was conducted on oncology
patients in a Peruvian public institution specializing in cancer. It was
selected because it allows for capturing a snapshot of health and mental health
problems in a representative sample, facilitating the rapid identification of
specific clinical needs and their association with quality of life
(Álvarez-Hernández & Delgado-De la Mora, 2015). In addition, the STROBE
guidelines were followed to ensure high-quality presentation of the study.
These guidelines consist of 22 elements, which are evaluated using a checklist
(Supplementary material 1).
Setting
The application of the instruments was performed for
two months (July and August 2018) by psychologists and psycho-pedagogues of the
Mental Health Unit of the "National Institute of Neoplastic Diseases"
(INEN, acronym in Spanish) who were previously trained in the administration of
psychometric tests. The tests were administered to each of the patients with
confirmed cancer separately, in areas of mental health, hospitalization, and
oncology outpatient clinics: Breast and mixed tumors, Gynecology, Medical Oncology,
Abdominal, Head and Neck, Urology, Thorax, Neuro-Oncology, and Orthopedics.
Individual administration preserves the independence of responses in
self-reported instruments like the SF-12 or HADS, strengthening internal
validity in cross-sectional designs by preventing social contagion or
suggestion effects among patients in shared rooms (Fowler, 2014; Gutiérrez, J P
et al., 2013). This approach accounts for confounding factors including
tiredness and environmental stress in hospital settings with a high oncological
burden.
Participants
The study population consisted of cancer cases
(11,894) registered in 2018 by the National Institute of Neoplastic Diseases (Ministerio de Salud, 2026). Due to the nature of the
sample, sampling was intentional and non-probabilistic. The inclusion criteria were
being over 18 years of age and having the ability to read and write. In
addition, patients who experienced physical discomfort during the tests and
individuals with cognitive disabilities that limited their comprehension and
ability to complete the instruments administered for this study were excluded.
The sample size was calculated a priori using Poisson
regression, assuming a small effect size (PR = 1.2), a significance level of
0.05, a statistical power of 90%, and an expected baseline prevalence of 43%
for the outcome in the unexposed group, based on previous literature (Suresh
& Chandrashekara, 2012). The calculation was performed using G*Power
version 3.1.9.3 (Faul et al., 2007, 2009), which resulted in a minimum required
sample size of 453 participants. However, due to potential non-response, inability,
and missing data, an additional 10% were surveyed, resulting in 500 responses
being recruited to ensure the minimum sample size.
Variables
Anxious symptoms: The Beck Anxiety Inventory (BAI) was used to assess anxiety symptoms.
Anxiety in BAI can be defined based on the criteria for anxiety described in
the DSM-III which are different from depressive symptoms (Beck et al., 1988).
Likewise, to differentiate anxious symptoms from depressive symptoms we can
define anxiety as fear, tension and apprehension usually associated with
anticipatory ideas of what may happen in the future and the activation of the
autonomic nervous system (Campo-Arias et al., 2014). The BAI is a 21-item
self-applied scale created by Beck et al. in 1988, that measures the severity
of anxiety symptoms in adults and adolescents in psychiatric populations (Beck
et al., 1988). The BAI is evaluated using a scale from 0 to 3 (0=Not at all,
1=Slightly, 2=Moderately, and 3=Severely), so the minimum score is 0 and the
maximum is 63 points. The questions refer to last week and the current moment;
administration can take approximately 15 minutes. It shows a high internal
consistency (α=0.93) and evidence of internal structure; it also has good
convergent validity with respect to other anxiety tests (Vizioli
& Pagano, 2020). Anxiety symptoms according to their scores are classified
as normal (0-9), low anxiety (10-18), moderate anxiety (19-29), and severe
anxiety (30-63) (Julian, 2011).
Perceived stress: The self-reported perceived stress scale with 10 items (PSS-10) was
used to assess perceived psychological stress. Perceived stress was defined as
the level of stress that the subject experiences as a function of objective
stressful events, coping processes, and personality factors, among others
(Cohen et al., 1983). PSS-10 is composed using a scale from 0 to 4 (0 = Never,
1 = Almost never, 2 = Occasionally, 3 = Often, and 4 = Very often). Construct
validity models of this test have been developed in different countries,
obtaining adequate scores for two-factor models, where the psychometric
properties of the PSS 10 were superior to those of the PSS 14 test (Campo-Arias
et al., 2009, 2014; Lee, 2013). We defined moderate and severe stress symptoms
using a cut-off of 14 and higher (Seedhom et al.,
2019).
Depression symptoms: The Beck Depression Inventory - Second Edition (BDI-II) was used to
assess depression symptoms and is defined in the specific subtype of depression
of the Diagnostic and Statistical Manual of Mental Disorders-Fourth Edition
(DSM-IV) (Beck et al., 1996). BDI-II is an inventory created by Beck for assessing
the severity of depressive symptoms in psychiatric patients and in adolescents
and adults (13 to 80 years) during the last 2 weeks (Beck et al., 1996). The
BDI-II consists of 21 items and 4 response alternatives ordered from least to
greatest severity. The response alternatives are valued from 0 to 3 (0=Not at
all, 1=Slightly, 2=Moderately, and 3=Severely), with a maximum score of 63 and
a minimum of 0.This inventory has high internal consistency (α = 0.94) and
evidence of internal structure supporting two factors (somatic-affective and
cognitive) in outpatients. Depressive symptoms, according to their scores, are
classified as mild depression (14-19), moderate depression (20-28), and severe
depression (29-63) (Farfán & Sánchez-Villena,
2019; Smarr & Keerfer, 2011).
Emotional distress: The Hospital Anxiety and Depression Scale (HADS) was used to assess
emotional distress. The National Comprehensive Cancer Network (NCCN) defines
distress as an unpleasant multi-determined emotional experience of a
psychological, social and spiritual nature that can interfere with the ability
to effectively cope with cancer, its physical symptoms and its treatment (Riba
et al., 2019). This emotional response ranges from common normal feelings of
vulnerability, sadness, and fear to problems that become disabling, such as
depression, anxiety, panic, social isolation, and spiritual crisis (Riba et
al., 2019). In this sense, emotional
distress is based on a transdiagnostic construct due to its ability to capture
shared emotional symptoms beyond specific categorical diagnoses, such as
anxiety and depression (Galindo Vázquez et al., 2015). In addition, the HADS
has proven to be an adequate and sensitive instrument to change both during the
disease and in response to psychotherapeutic treatment and psychopharmacological
intervention (Fernández-de-las-Peñas et al., 2022;
Nikolovski et al., 2024).
Unlike disorder-specific instruments (BDI-II, BAI),
the HADS captures a broader, transdiagnostic emotional burden characterized by
generalized psychological distress and impaired emotional coping, while
minimizing the overlap of somatic symptoms common in cancer populations
(Bjelland et al., 2002). Therefore, depressive symptoms, anxiety, and perceived
stress were considered specific indicators of the disorder, while emotional
distress was treated as a higher-order transdiagnostic
construct.
The HADS was created by Zigmond and Snaith in 1983
(Zigmond & Snaith, 1983), and translated from
English into Spanish by Tejero et al. (1986). This instrument consists of 14
items and 2 subscales (each of 7 items) that assess symptoms of anxiety and
depression at a cognitive and discomfort emotional level in patients with
somatic illnesses during the last week. The HADS has a Likert-type response
option from 0 to 3, so the scores on each subscale range from 0 to 21. For both
scales, if they exceed 8 points, it is considered "case" and scores
higher than 11 points are a "probable case" (Zigmond & Snaith,
1983). However, a meta-analysis study suggests that cancer patients consider a
cut-off point scores greater than 15 for the total HADS (sensitivity 0.87;
specificity 0.88) (Vodermaier & Millman,
2011).The psychometric properties of HADS show high internal consistency (Alpha
anxiety = 0.68-0.93 and Alpha depression = 0.67-0.90) in systematic reviews of
hospital and chronic populations (Terol-Cantero et al., 2015).
Quality of Life: The quality of life was assessed using the short form 12 questionnaire
(SF-12). The SF-12 assesses eight domains of quality of life, four related to
physical health (General Health, Physical Functioning, Role Physical, and Body
Pain) and four related to mental health (Vitality, Social Functioning, Role
Emotional, and Mental Health) (Huo et al., 2018). The response options to the
SF-12 items are dichotomous (yes or no) and Likert-type. Response options are
scored, weighed, and summed to produce physical and
mental component scores ranging from 0 to 100, with higher scores indicating
better quality of life in that domain. The scoring of the SF-12 was performed
using the STATA package developed by Bruun (Bruun, 2021). The SF-12 has high
internal consistency with Cronbach's alphas above 0.80 and a test-retest
correlation of 0.78-0.89 for the general and Spanish populations; it also has
convergent and discriminant validity consistent with other tests (Schmidt et
al., 2012).
Covariates
Other sociodemographic variables assessed in the study
included: sex (women/men); age grouped
into four groups of approximately 15 years each, following the life cycle
scheme from age 17 onwards (young people: 17 to 29, young adults: 30 to 44,
adults: 45 to 59, and older adults: 60 and over) (Instituto Nacional de Estadística e Informática, 2014);
type of care (Outpatient clinic, those who have scheduled appointments such as
chemotherapy, blood tests, radiation therapy, among others; Outpatient
consultation, for patients who are referred for medical or surgical procedures
in this specialty; and Hospitalization, one that requires the patient to be
admitted) (EsSalud, 2020); Civil status (With a
current couple, Separated or Widower, and Single); Educational years (primary
education [At least 6 years old], secondary education [7 to 11 years], and
superior education [12 to more years]); and laboral
status (with work and without work). Also, the variable types of cancer
(focused/unfocused) are considered, prioritizing the cancers with the highest
mortality worldwide with sufficient frequency (lung cancer [bronchi, lungs, and
trachea], colorectal cancer, gastric cancer, breast cancer, cervical cancer,
and others focal type of cancer) (Cancer (IARC) & The International Agency
for Research on Cancer, 2022). In addition, the clinical stage variable (early
stages [0, I], advanced stages [II, III, IV], and there is no record)
(Instituto Nacional del Cáncer, 2015) and comorbidity
of mental health problems (number of mental health problems that participants
have) were included. Given the high degree of conceptual and empirical
relationship between specific mental health symptoms and the comorbidity
covariate, separate models were analyzed for the latter to avoid multicollinearity
and overfitting.
Statistical analysis
Characteristics of the participants: In the descriptive analysis, we report the frequencies and percentages
of all sociodemographic variables and the prevalence of depression, anxiety,
and stress symptoms. Also, an analysis of the prevalence of moderate to severe
symptoms of mental health problems (anxiety, depression, stress, and emotional
distress) and quality of life in relation to clinical stage and the types of
cancer with the highest overall mortality was performed.
Validity of the models: Based on the conceptual framework (Figure 1), three complementary
regression models were specified to assess the association between mental
health symptoms and quality of life, minimizing multicollinearity, which was
evaluated using variance inflation factors (VIF). In the first model, specific
mental health symptoms such as depression, anxiety, and perceived stress were
considered independent predictors; the second, transdiagnostic model assessed
emotional distress as a higher-order construct; finally, the third model
examined comorbidity as an indicator of cumulative symptom burden. Covariates
for multivariable models were chosen based on statistical criteria, clinical
plausibility, previous empirical data, and their possible confounding effects.
All adjusted models showed VIF values below 5. Variables were first examined in
crude models and subsequently entered simultaneously into multivariable models
if they met statistical criteria (p < 0.05), had clinical plausibility, or were
supported by previous literature as potential confounders.
Figure 1. Integrative Conceptual Model
Mental health and quality of life outcomes: Quality of life (mental and physical components) was considered the
outcome variable, while depressive symptoms, anxiety, perceived stress, and
emotional distress were treated as exposure variables. Mental health variables
included anxiety, stress, depression, and emotional distress. These were
dichotomized: mild perceived stress (score of 0 to 13) and moderate to severe
perceived stress (score of 14 to 40); normal to mild depressive symptoms (score
of 0 to 18) and moderate to severe depressive symptoms (score of 19 and above);
normal to mild anxiety symptoms (score of 0 to 18) and moderate to severe
symptoms (score of 19 and above); and absence of emotional distress (score of 0
to 10) and probable cases of emotional distress (score of 11 or above), based on
the criteria of Zigmond and Snaith, considering the scores with greater
clinical specificity (Zigmond & Snaith, 1983). On the other hand, quality
of life includes two dimensions: mental quality of life and physical quality of
life. The scores obtained were divided into tertiles, which were then
dichotomized into low-medium and high (high quality of life was used as the
reference category to facilitate interpretation), since there are no universal
clinical cut-off points (Bruun, 2021). We consider moderate to severe symptoms
and low-medium quality of life to be clinically significant for cancer patients
who need mental health support (outcomes). A sub-analysis of the comorbidity of
mental health problems was performed for mental quality of life and physical quality
of life.
In addition, generalized linear models with the
Poisson family were used to calculate crude (crPR)
and adjusted (aPR) prevalence ratios and their 95%
confidence intervals (95% CI) between each covariate and the dichotomous
outcomes of mental health and quality of life. To determine the covariates that
would be included in the adjusted model, the criterion used was that they had a
p-value of less than 0.05 in the crude model, to be added to the multivariate
model. By including interaction terms in Poisson regression models with robust
variance, potential impact modification by sex was investigated in the
relationship between emotional distress and both physical and mental quality of
life. Since the interaction terms were not statistically significant (p >
0.10), neither model showed any indication of modification of effect by sex.
The amount and direction of the relationship between emotional distress and
poorer physical and mental quality of life were consistent for both sexes. In
addition, sensitivity analyses were performed using log-binomial regression
models to estimate prevalence ratios. Although convergence problems and
unacceptable predicted values were observed in some specifications, the results
were consistent in direction and magnitude with those obtained using robust
Poisson regression. Therefore, robust Poisson regression was retained as the
primary analytical approach due to its numerical stability and widespread use
in cross-sectional studies, while log-binomial models were used only for sensitivity
analyses.
Topics of Ethics
The protocol was approved by the INEN Research Ethics
Committee and the Research Review Committee (N°239-2018-CIE/INEN). Participants
were invited to participate in research according to conventional ethical
requirements. Subsequently, signed the written informed consent, and were
provided with the questionnaire, which consisted of socio-demographic questions
and psychometric tests.
RESULTS
Characteristics of the participants
Initially, 500 participants were evaluated, but those
who lacked information on the outcome of interest (n = 27, 5.3%) or who were
foreigners (n = 8, 1.6%) were eliminated. There were no missing values. After
filtering, a total of 465 participants were obtained, exceeding the minimum
sample size required (453). Most participants were women (75.7%), aged between
17 and 84 (mean = 45.9; SD = 14.4), were currently in a romantic relationship
(48.2%), had focal cancer (77.6%), were in an advanced stage of cancer (38.3%),
and were unemployed (78.3%), mostly homemakers. The characteristics of the
participants are presented in Table 1. In addition, the prevalence of anxiety
symptoms of 27.5%, depressive symptoms of 20.4%, moderate-severe stress
symptoms of 83.2%, and emotional distress of 57.41% was identified. Our study
reveals that 87.1% of participants had some mental health problems.
Table 1. Characteristics of the
participants (n=465).
|
|
|
n |
% |
|
Sex |
Men |
113 |
24.3% |
|
Women |
352 |
75.7% |
|
|
Age |
17 to 29 |
72 |
15.5% |
|
30 to 44 |
141 |
30.3% |
|
|
45 to 59 |
164 |
35.3% |
|
|
60 to more |
88 |
18.9% |
|
|
Type of care |
Outpatient clinic |
185 |
39.8% |
|
Outpatient |
154 |
33.1% |
|
|
Hospitalization |
126 |
27.1% |
|
|
Civil status |
With current couple |
224 |
48.2% |
|
Separated or Widower |
74 |
15.9% |
|
|
Single |
167 |
35.9% |
|
|
Educational years |
At least 6 years old |
78 |
16.8% |
|
7 to 11 years |
214 |
46.0% |
|
|
12 to more |
173 |
37.2% |
|
|
Laboral status |
With work |
101 |
21.7% |
|
Without work |
364 |
78.3% |
|
|
Clinical Stage |
Early stage |
187 |
40.2% |
|
Advanced stage |
178 |
38.3% |
|
|
No stage |
100 |
21.5% |
|
|
Type of cancer |
Unfocused |
104 |
22.4% |
|
Focused |
361 |
77.6% |
|
|
Focal type of cancer |
Colorrectal cancer |
15 |
3.2% |
|
Breast cancer |
137 |
29.5% |
|
|
Cervical cancer |
69 |
14.8% |
|
|
Gastric cancer |
19 |
4.1% |
|
|
Lung cancer |
14 |
3.0% |
|
|
Otros |
211 |
45.4% |
|
|
Anxious symptoms |
None to Leve |
337 |
72.5% |
|
Moderate to Severe |
128 |
27.5% |
|
|
Depression symptoms |
None to Leve |
370 |
79.6% |
|
Moderate to Severe |
95 |
20.4% |
|
|
Moderate-severe stress |
Leve |
78 |
16.8% |
|
Moderate to Severe |
387 |
83.2% |
|
|
Emotional Distress |
No |
198 |
42.6% |
|
Yes |
267 |
57.4% |
|
|
Comorbidity of mental health
problems |
None |
60 |
12.9% |
|
One mental health problem |
143 |
30.8% |
|
|
|
Two or more mental health
problems |
262 |
56.3% |
Figure 2 shows the prevalence of factors associated
with mental health according to the type of cancer-focused according to gender.
The prevalence of symptoms of anxiety (33.33%) and moderate to severe
depression (33.33%) is higher in women with gastric cancer compared to men.
Likewise, the prevalence of signs of stress and emotional distress are higher
in women with colorectal cancer (88.89%), and other types of focused cancer
(77.78%). Regarding low to medium quality of life, the prevalence is higher in
women with lung cancer (100%) and/or other focal cancers (67.21%). Finally,
women with cervical cancer have a higher prevalence of stress (94.20%) and a
low to medium mental (81.16%) and physical (71.01%) quality of life. A
sub-analysis of the prevalence of mental health and quality of life outcomes by
clinical stage can be found in Supplementary Material 2.
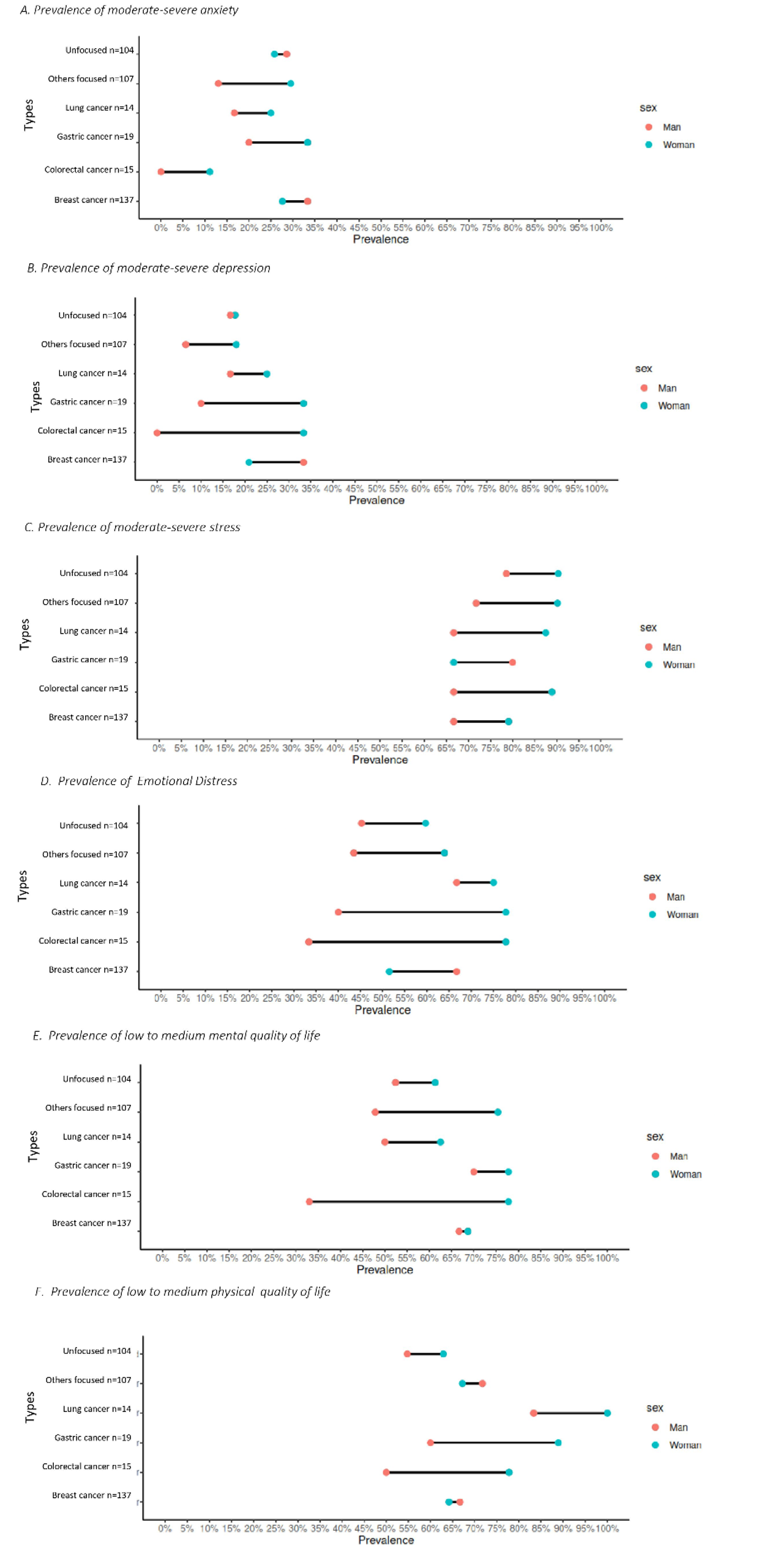
Figure 2. Prevalence of mental health and quality of life
outcomes according to type of cancer and gender.
Mental health and Quality of Life
In the regression model, participants with cancer who
had at least 12 years of education (college or higher technical education) were
33% less likely to have emotional distress, compared to those with basic
education (aPR= 0.67, 95%CI=0.48-0.94) (see Table 2).
It should be noted that neither cancer (unfocused and focused) nor clinical
stage (early stage, advanced stage, and no stage) presented significant values
for any of the mental health problems (anxious symptoms, stress, depression
symptoms, and emotional distress).
Table 2. Regression model for associated
factors to mental health (n=465).
|
Anxious symptoms |
Moderate-severe stress |
Depression symptoms |
Emotional Distress |
|||
|
|
|
rPR (95% CI) |
rPR (95% CI) |
rPR (95% CI) |
rPR (95% CI) |
aPR(95%CI)* |
|
Sex |
Men |
Ref. |
Ref. |
Ref. |
Ref. |
Ref. |
|
|
Women |
1.55 (0.98-2.45) |
1.16 (0.91-1.47) |
2.02 (1.13-3.64) |
1.36 (1.00-1.84) |
1.34 (0.98-1.81) |
|
Age |
17 to 29 |
Ref. |
Ref. |
Ref. |
Ref. |
- |
|
30 to 44 |
1.25 (0.72-2.16) |
0.93 (0.68-1.26) |
1.26 (0.69-2.29) |
1.07 (0.74-1.56) |
- |
|
|
45 to 59 |
1.24 (0.73-2.13) |
0.94 (0.70-1.26) |
0.82 (0.44-1.53) |
1.02 (0.70-1.48) |
- |
|
|
|
60 to more |
0.68 (0.34-1.35) |
0.89 (0.64-1.26) |
0.82 (0.40-1.67) |
1.02 (0.67-1.55) |
- |
|
Type of care |
Outpatient clinic |
Ref. |
Ref. |
Ref. |
Ref. |
- |
|
Outpatient |
1.04 (0.70-1.55) |
0.98 (0.77-1.24) |
1.57 (0.99-2.46) |
1.25 (0.95-1.65) |
- |
|
|
|
Hospitalization |
0.88 (0.56-1.37) |
1.00 (0.78-1.29) |
0.85 (0.48-1.49) |
1.04 (0.77-1.42) |
- |
|
Civil status |
With current couple |
Ref. |
Ref. |
Ref. |
Ref. |
- |
|
Separated or Widower |
0.90 (0.53-1.55) |
0.98 (0.73-1.31) |
0.76 (0.39-1.47) |
0.93 (0.65-1.33) |
- |
|
|
Single |
1.27 (0.88-1.84) |
1.02 (0.82-1.27) |
1.22 (0.79-1.87) |
1.00 (0.77-1.30) |
- |
|
|
Educational years |
At least 6 years old |
Ref. |
Ref. |
Ref. |
Ref. |
Ref. |
|
7 to 11 years |
0.89 (0.56-1.42) |
0.87 (0.66-1.15) |
1.05 (0.61-1.82) |
0.78 (0.57-1.06) |
0.80 (0.59-1.10) |
|
|
12 to more |
0.76 (0.46-1.24) |
0.90 (0.67-1.19) |
0.77 (0.42-1.40) |
0.66 (0.47-0.92) |
0.67 (0.48-0.94) |
|
|
Laboral status |
Without work |
Ref. |
Ref. |
Ref. |
Ref. |
- |
|
With work |
0.71 (0.44-1.13) |
0.92 (0.72-1.18) |
0.96 (0.59-1.57) |
0.73 (0.53-1.01) |
- |
|
|
Clinical stage |
Early stage |
Ref. |
Ref. |
Ref |
Ref. |
- |
|
|
Advance stage |
0.83 (0.56-1.23) |
1.04 (0.83-1.31) |
0.81 (0.52-1.27) |
0.85 (0.65-1.11) |
- |
|
|
No stage |
0.85 (0.54-1.36) |
1.07 (0.82-1.39) |
0.72 (0.41-1.26) |
0.86 (0.62.1.18) |
- |
|
Type of cancer |
Unfocused |
Ref. |
Ref. |
Ref. |
Ref. |
- |
|
Focused |
1.03 (0.68-1.56) |
0.96 (0.76-1.22) |
1.23 (0.74-2.06) |
1.09 (0.81-1.46) |
- |
|
Note: rPR
= Raw prevalence ratio. aPR = Adjusted prevalence
ratio. 95% CI = 95% confidence interval. *Model adjusted for Emotional Distress
ajusted by sex and educational years. Values in bold are significant
(p<0.05).
Cancer patients with a low or average quality of
mental life are twice as likely to present signs of moderate or severe stress (aPR=2.30, 95%CI=1.42-3.69). Similarly, they are 46% more
likely to present symptoms of emotional distress (aPR=1.46,
95%CI=1.10-1.96). In addition, cancer patients with a low and medium physical
quality of life are 49% more likely to suffer emotional distress (RPa=1.49, 95%CI=1.18-1.89) than those without emotional
distress (see Table 3). It should be noted that neither cancer type nor cancer
stage presented significant values for any mental health problems (anxious
symptoms, stress, depression symptoms, and emotional distress). In addition, it
should be noted that neither cancer (unfocused and focused) nor clinical stage
(early stage, advanced stage, and no stage) presented significant values for
mental quality of life or physical quality of life (Supplementary material 3 y
4).
Table 3. Regression model for
associated dimensions to quality of life outcomes
(n=465).
|
Mental quality
of life (low-middle) |
Quality of life
Physical (low-middle) |
||||
|
|
|
rPR (95% CI) |
aPR (95%CI)* |
rPR (95% CI) |
aPR (95%CI)** |
|
Sex |
Men |
Ref. |
Ref. |
Ref. |
- |
|
|
Women |
1.37 (1.03-1.81) |
1.17 (0.88-1.56) |
1.06 (0.82-1.38) |
- |
|
Age |
17 to 29 |
Ref. |
- |
Ref. |
- |
|
30 to 44 |
1.03 (0.73-1.46) |
- |
1.39 (0.96-2.03) |
- |
|
|
45 to 59 |
1.02 (0.72-1.42) |
- |
1.33 (0.92-1.93) |
- |
|
|
|
60 to more |
0.92 (0.62-1.36) |
- |
1.33 (0.88-2.00) |
- |
|
Type of care |
Outpatient clinic |
Ref. |
- |
Ref. |
- |
|
Outpatient |
0.97 (0.75-1.26) |
- |
0.99 (0.76-1.29) |
- |
|
|
|
Hospitalization |
0.99 (0.75-1.30) |
- |
1.05 (0.80-1.38) |
- |
|
Civil status |
With current couple |
Ref. |
- |
Ref. |
- |
|
Separated or Widower |
0.97 (0.69-1.35) |
- |
0.91(0.66-1.26) |
- |
|
|
Single |
1.12 (0.88-1.42) |
- |
0.92 (0.72-1.18) |
- |
|
|
Educational years |
At least 6 years old |
Ref. |
- |
Ref. |
- |
|
7 to 11 years |
0.86 (0.63-1.16) |
- |
0.87 (0.64-1.19) |
- |
|
|
12 to more |
0.82 (0.60-1.12) |
- |
0.88 (0.64-1.21) |
- |
|
|
Laboral status |
Without work |
Ref. |
- |
Ref. |
- |
|
With work |
0.85 (0.64-1.12) |
- |
0.92 (0.70-1.21) |
- |
|
|
Clinical stage |
Early stage |
Ref. |
- |
Ref. |
- |
|
|
Advance stage |
1.06 (0.83-1.36) |
- |
1.15(0.89-1.47) |
- |
|
|
No stage |
0.96 (0.71-1.30) |
- |
1.00 (0.74-1.36) |
- |
|
Type of cancer |
Unfocused |
Ref. |
- |
Ref. |
- |
|
Focused |
1.20 (0.91-1.59) |
- |
1.15 (0.87-1.52) |
- |
|
|
Anxious symptoms |
No |
Ref. |
Ref. |
Ref. |
Ref. |
|
|
Yes |
1.57(1.25-1.98) |
1.10 (0.83-1.43) |
1.27 (1.00-1.61) |
1.07 (0.82-1.39) |
|
Depression symptoms |
No |
Ref. |
Ref |
Ref. |
- |
|
|
Yes |
1.52(1.19-1.95) |
1.11 (0.83-1.48) |
1.29 (0.99-1.67) |
- |
|
Moderate-severe stress |
No |
Ref. |
Ref. |
Ref. |
- |
|
|
Yes |
2.92(1.86-4.60) |
2.30 (1.42-3.69) |
1.36 (0.98-1.90) |
- |
|
Emotional Distress |
No |
Ref |
Ref. |
Ref. |
Ref. |
|
|
Yes |
1.90(1.48-2.44) |
1.46 (1.10-1.96) |
1.49 (1.18-1.89) |
1.45(1.12-1.88) |
Note: rPR
= Raw prevalence ratio. aPR = Adjusted prevalence
ratio. 95% CI = 95% confidence interval. *Multiple Poisson regression for
Mental quality of life ajusted by sex, Anxious
symptoms, Depression symptoms, Stress, and Emotional Distress. **Multiple
Poisson regression for Quality of life Physical ajusted
by Anxious symptoms and Emotional Distress. Values in bold were significant (p<0.05).
The comorbidity sub-analysis identified that as the
number of comorbid mental health problems increases, the probability of having
a low mental quality of life and physical quality of life increases (see Table
4). Having at least one mental health problem increases the probability of
having a poor mental quality of life (aPR=2.55,
95%CI=1.38-4.69) compared to having no mental health problem. Also, having two
or more mental health problems increases the probability of having a poor
physical quality of life (aPR=1.68, 95%CI=1.13-2.50)
compared to having one or no mental health problem (Supplementary material 5).
Table 4. Regression model of quality of life outcomes by comorbidity of mental health
problems (n=465).
|
|
|
Mental quality
of life (low-middle) |
Quality of life
Physical (low-middle) |
||
|
rPR (95% CI) |
aPR (95%CI)* |
rPR (95% CI) |
aPR (95%CI)* |
||
|
Comorbidity of mental health
problems |
None |
Ref. |
Ref. |
Ref. |
- |
|
One |
2.59 (1.41-4.76) |
2.55 (1.38-4.69) |
1.17 (0.76 - 1.80) |
1.17 (0.76-1.81) |
|
|
Two or more |
4.27 (2.39-7.64) |
4.13 (2.30-7.41) |
1.67 (1.12- 2.48) |
1.68 (1.13-2.50) |
|
Note: rPR
= Raw prevalence ratio. aPR = Adjusted prevalence
ratio. 95% CI = 95% confidence interval. *Multiple Poisson regression ajusted by sex. Values in bold were significant
(p<0.05).
DISCUSSION
Main findings and significance of the results
The findings show that mental health symptoms, whether
assessed specifically or from a transdiagnostic perspective, are consistently
associated with lower physical and mental quality of life in Peruvian cancer
patients. Emotional distress and perceived stress act as the main predictors of
low well-being, reinforcing the need for a comprehensive and timely assessment
of psychological distress beyond the primary cancer diagnosis.
The prevalence of depression (20.4%) and anxiety
(27.5%) found is consistent with meta-analyses in low- and middle-income
countries. However, the prevalence of stress (83.2%) and emotional distress
(57.4%) was higher than reported in international studies on breast or ovarian
cancer (Walker et al., 2021). Unlike some studies that suggest variations
depending on the type of cancer, our study found no significant association
between the type or stage of cancer and mental health problems, which is
consistent with literature suggesting that self-awareness of chronic illness is
a more determining factor than the specific location of the cancer (Martínez
López et al., 2017; Ploos van Amstel et al., 2015;
Goebel et al., 2011; Ludwigson et al., 2020).
Empirical evidence remains contradictory regarding
differences in mental health outcomes between different types and stages of
cancer. While some studies suggest that cancer patients are heterogeneous and
experience different psychological problems depending on the type and stage of
the disease (Abbas et al., 2021; Aquil et al., 2021; Walker et al., 2021; Wen
et al., 2019), others have found no significant differences between different
types of cancer (Li et al., 2018). These inconsistencies can be explained by
variations in sociocultural contexts at the time of diagnosis, the limited
sample sizes in many studies, and the use of different assessment instruments.
Furthermore, it is plausible that factors such as awareness of having a chronic
illness play a more important role in the development of mental health problems
than the specific anatomical location of cancer. Previous research has shown
that awareness of having a chronic illness is associated with an increased risk
of psychological distress and mental health disorders (Ravindran et al., 2019;
Yeh et al., 2021).
From a transdiagnostic approach, the results support
that emotional distress is the most important component in predicting poor
mental and physical quality of life. Several cross-sectional studies have
previously identified an inverse relationship between quality of life, stress,
and emotional distress in cancer patients (Prapa et
al., 2021; Ravindran et al., 2019; Yeh et al., 2021).The use of three
complementary models showed that symptom comorbidity matters more than isolated
symptoms; having two or more mental health problems dramatically increases the
likelihood of poor physical and mental quality of life (Table 4 and
Supplementary Material 2). One of these models is the hierarchical taxonomy of
psychopathology, which posits that emotional distress and perceived stress are
the highest-order components within the range of emotional disorders (Kotov et
al., 2017). This cumulative burden
reflects a clinical severity that requires interventions addressing shared core
vulnerabilities.
It was identified that gender is a risk factor for the
presence of depressive symptoms. There is ample evidence that gender is a
predictor of the presence of depressive symptoms. A global meta-analysis
reported that women have a prevalence of 31%, compared to a prevalence of 26%
in men (Mejareh et al., 2021). This may be due to a
variety of biological and hormonal factors in women that increase the risk of
having intense emotional symptoms (Grossman & Wood, 1993). However, as
studies have shown that men had higher symptoms (Park & Kim, 2021), this
may be due to social patterns. In a society with traditional gender norms
masculinity involves hiding concerns and not expressing feelings of
vulnerability (Fish et al., 2015). The cancer diagnosis can undermine
perceptions of masculinity, coupled with poor control of the effects of the
disease, leading to further deterioration of their health in the long term
(Park & Kim, 2022). While women are better at identifying emotions (Barabanschikov & Suvorova, 2021; Gordillo-León et al.,
2021). So, they would be better able to identify symptoms of sadness and
emotional distress and therefore present a higher rate of positive cases.
A higher level of education was identified as a
protective factor for emotional distress symptoms. The literature exposes
contrary positions, as one systematic review reported that educational level
does not predict long-term emotional distress (Cook et al., 2018). Other
studies using the distress thermometer instrument, which has acceptable
diagnostic performance for measuring emotional distress (Priede et al., 2014),
found that lower educational levels are associated with greater distress and
patients with lower educational levels are more likely to develop emotional
distress (Duan et al., 2021; Kim et al., 2011; Merckaert
et al., 2005; Wang et al., 2018). Another possible explanation is that
educational level is a predictor of income level in Peru, and there is evidence
that a high-income level is a predictor of the presence of emotional problems,
such as depressive symptoms (Villarreal-Zegarra et al., 2020).
Relevance in public health
Quality of life is a relevant outcome within the
course of cancer disease (Cao et al., 2017; Fernández de Larrea-Baz et al.,
2020; Hu et al., 2021; Li et al., 2018; Lu et al., 2019; Mace et al., 2021;
Ngan et al., 2021; Nipp et al., 2017; Ravindran et al., 2019), so healthcare
systems should develop interventions and strategies to care for the quality of
life of cancer patients. Our study helps to prioritize which mental health
problems are associated with poor quality of life (i.e., stress and emotional
distress). Therefore, we suggest that future studies focused on mental health
problems could be based on the transdiagnostic approach and offer further
support for these interventions in cancer patients. Interventions based on a
transdiagnostic approach have been shown to have several public health
advantages by grouping people with common emotional distress symptoms,
associated with a negative impact on their quality of life (González-Blanch et
al., 2018). Also, the transdiagnostic approach allows for a low-cost,
short-duration implementation that will optimize mental health care by
improving the quality of life for a wide range of cancer patients (Dalgleish et
al., 2020; González-Blanch et al., 2018).
Our study also serves as an input for cancer insurance
policies in low- and middle-income countries, as it reveals a high prevalence
and comorbidity of mental health problems. Consequently, it would allow them to
plan actions to promote a culture of prevention, early detection and treatment,
restoring quality of life to patients. Additionally, the establishment of
psychological and psychiatric care plans for cancer patients within their
policies is suggested.
In Peru, since 2015, the Ministry of Health and INEN
have implemented a budgetary program for the control and promotion of mental
health that focuses on the application of psychological support strategies for
cancer patients (Instituto Nacional de Enfermedades Neoplásicas, 2017; Ley Nacional del Cáncer,
2021). During the COVID-19 pandemic, mental health support, and self-care
strategies for cancer patients were strengthened. One of the main objectives of
the program is the improvement of the quality of life through a psychosocial
support team (Instituto Nacional de Enfermedades Neoplásicas, 2020; Ministerio de
Salud, 2020). Our results support the need to further strengthen this type of
health strategy.
Strengths and limitations
This study presents some methodological limitations.
First, this study has a cross-sectional design that consists of making a single
measurement over some time and analyzing the relationship between variables
(Cvetkovic-Vega et al., 2021), so it is not possible to make statements about
the causal relationship between variables. Nevertheless, these results are
important because they show what associated factors are important for the
mental health and quality of life of cancer patients. Second, cancer patients
are influenced by their family and social environment, which can increase their
risk of mental health problems (Yeh et al., 2021), but it is not possible to
assess patients and relatives, so this could introduce a measurement bias.
Third, the results are not generalizable to other cancer populations in Peru,
because our study is non-probabilistic. Fourth, the sample size is small, so
regression analyses cannot be performed for each cancer diagnosis (i.e.,
gastric cancer, cervical cancer, or others).
Conclusions
In relation to the main objective, it was found that
the main factors associated with physical quality of life and mental quality of
life are emotional distress and stress. On the other hand, it was observed that
a high level of education was a factor associated with emotional distress, but
the type of cancer and clinical stage did not obtain significant results.
Finally, it was found that the greater the comorbidity of mental health
problems, the greater the association with a lower quality of life (physical
and mental).
Based on the findings, there is a need for oncology
services to incorporate routine mental health screening protocols that assess
the presence of comorbid psychological symptoms, which would allow for the
early identification of patients at greater risk of deterioration in their
quality of life. In this regard, it is suggested that interventions with a
transdiagnostic approach be implemented, aimed at reducing emotional distress
and stress in oncology patients, considering the comorbidity identified among the
symptoms evaluated. Likewise, for future research, it is recommended to use
structural equation modelling (SEM) to explore the complex relationships
between depression, anxiety, stress, emotional distress, and quality of life,
facilitating the assessment of latent constructs and possible mediating
mechanisms.
ORCID
Sofía C. Malaquias-Obregon: https://orcid.org/0000-0002-0049-7470
David Villarreal-Zegarra: https://orcid.org/0000-0002-2222-4764
Jackeline García-Serna: https://orcid.org/0000-0001-9260-1505
Milagros Cabrera-Alva: https://orcid.org/0000-0002-7345-878X
Anthony Copez-Lonzoy: https://orcid.org/0000-0003-4761-4272
Ana L. Vilela-Estrada: http://orcid.org/0000-0001-5647-465X
AUTHORS’ CONTRIBUTION
Sofía C.
Malaquias-Obregon: Formal Analysis, Investigation, Validation, Writing –
Original version, Approval of the final version.
David
Villarreal-Zegarra: Formal Analysis, Methodology, Supervision, Validation,
Writing – Original version, Approval of the final version.
Jackeline
García-Serna: Investigation, Validation, Writing – Original version, Approval
of the final version.
Milagros Cabrera-Alva:
Investigation, Validation, Writing – Original version, Approval of the final
version.
Ana L. Vilela-Estrada:
Conceptualization, Data Curation, Investigation, Methodology, Validation,
Writing – Review & Editing, Approval of the final version.
Anthony Copez-Lonzoy: Conceptualization, Methodology, Validation, Writing
– Review & Editing, Approval of the final version.
FUNDING
SOURCE
This research did not
receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICT
OF INTEREST
The authors report no conflict of interest
when conducting the study, analyzing the data, or writing the manuscript.
ACKNOWLEDGMENTS
The authors thank the support of the
members of the Oncological Mental Health Functional Team; evaluating clinical
psychologists: Rosa Argüelles Torres, Giovanna Galarza Torres, Flor Arrunátegui Reyes, Hernán Bernedo
Del Carpio, Antonio Conso Machuca, Oscar Villanueva Cortés, Sarita Angulo
Rubio, Yvo Fernández Montoro, the Psychology Interns team period 2018 and all
patients of the INEN that responded to the evaluation. Also, to Daniel Rivas
for the linguistic revision of the article.
REVIEW
PROCESS
This study has been reviewed by two external reviewers in double-blind
mode by Carlos Narváez Gaitán and another reviewer. The editor in charge was Renzo
Rivera. The review process is included as supplementary material 6.
DATA AVAILABILITY
STATEMENT
The authors
provide the database at https://doi.org/10.6084/m9.figshare.28046711.
DECLARATION OF THE USE OF GENERATIVE ARTIFICIAL INTELLIGENCE
We used DeepL to translate specific sections
of the manuscript and Grammarly to improve the wording of certain sections. The
final version of the manuscript was reviewed and approved by all authors.
DISCLAIMER
The authors are responsible for all statements made in this article.
REFERENCES
Abbas, Q., Kanwal, U., Khan, M. U., Saeed,
W., & Shahzadi, M. (2021). Role of religiosity, optimism, demographic
characteristics and mental health problems among cancer patients. Journal of
the Pakistan Medical Association, 71(3), 859–862. https://doi.org/10.47391/JPMA.1013
Adekunle, A. O., Abiodun, A., Akingbola,
A., Tundealao, S., Anyagwa,
O. E., Mobolaji, A. A., Badejo, A., Daniel, O. D., Oduntan,
O., Victor, O. O., Bale, B. I., Oluwatoyin, O. I., Folajimi, A., & Chuku,
J. (2025). Financial toxicity of surgical cancer treatment in LMICs:
Implications for patients and health systems. Journal of Cancer Policy, 45,
100616. https://doi.org/10.1016/j.jcpo.2025.100616
Ahmad, W., Iqbal, S. M., & Jamil, Q.
(2025). Exploring financial toxicity and its impact on health-related quality
of life of cancer patients. Supportive Care in Cancer, 33(4), 294. https://doi.org/10.1007/s00520-025-09357-1
Álvarez-Hernández, G., & Delgado-De la Mora, J.
(2015). Diseño de estudios epidemiológicos. I. El estudio transversal: Tomando
una fotografía de la salud y la enfermedad. Boletín Clínico del Hospital
Infantil del Estado de Sonora, 32(1), 26–34.
American Cancer Society. (2022).
Cancer staging: Understanding
your diagnosis. https://www.cancer.org/treatment/understanding-your-diagnosis/staging.html
Antoni, M. H., & Dhabhar,
F. S. (2019). The impact of
psychosocial stress and stress management on immune responses in patients with
cancer. Cancer, 125(9), 1417–1431. https://doi.org/10.1002/cncr.31943
Aquil, A., El Kherchi, O., El Azmaoui, N., Mouallif, M.,
Guerroumi, M., Benider, A., & Elgot,
A. (2021). Predictors of
mental health disorders in women with breast and gynecological cancer after
radical surgery: A cross-sectional study. Annals of Medicine and Surgery, 65,
Article 102278. https://doi.org/10.1016/j.amsu.2021.102278
Barabanschikov, V. A., & Suvorova, E.
V. (2021). Gender
differences in the recognition of emotional states. Psychological Science
and Education, 26(6), 107–116. https://doi.org/10.17759/PSE.2021260608
Beck, A. T., Epstein, N., Brown, G., &
Steer, R. A. (1988). An inventory for measuring clinical anxiety: Psychometric
properties. Journal of Consulting and Clinical Psychology, 56(6),
893–897. https://doi.org/10.1037/0022-006X.56.6.893
Beck, A. T., Steer, R. A., & Brown, G.
K. (1996). BDI-II, Beck Depression Inventory: Manual. Psychological
Corporation.
Berger, A. M., Abernethy, A. P., Atkinson,
A., Barsevick, A. M., Breitbart, W. S., Cella, D., Cimprich, B., Cleeland, C., Eisenberger, M. A., Escalante,
C. P., Jacobsen, P. B., Kaldor, P., Ligibel, J. A.,
Murphy, B. A., O’Connor, T., Pirl, W. F., Rodler, E., Rugo, H. S., Thomas, J.,
& Wagner, L. I. (2010). Cancer-related fatigue. Journal of the National
Comprehensive Cancer Network, 8(8), 904–931. https://doi.org/10.6004/jnccn.2010.0067
Bergerot, C. D., Bergerot, P. G.,
Philip, E. J., Ferrari, R., Peixoto, R. M., Crane, T. E., Schmitz, K. H., &
Soto-Perez-de-Celis, E. (2024). Enhancing cancer supportive care:
Integrating psychosocial support, nutrition, and physical activity using
telehealth solutions. JCO Global Oncology, 10. https://doi.org/10.1200/GO-24-00333
Bjelland, I., Dahl, A. A., Haug, T. T.,
& Neckelmann, D. (2002). The validity of the Hospital Anxiety and
Depression Scale: An updated literature review. Journal of Psychosomatic
Research, 52(2), 69–77. https://doi.org/10.1016/S0022-3999(01)00296-3
Bruun, N. H. (2021). SF12: Stata module
to validate SF-12 input and calculate SF-12 version 2 t scores [Software]. https://econpapers.repec.org/software/bocbocode/s458125.htm
Bustamante, R. I., Vela-Ruiz, J. M. ,
Paredes-Olivares, O., & Carreño-Escobedo, R. A. (2022). Gaps in access to oncological treatment in
a reference health hospital in the south of Lima in 2019. Revista de la Facultad de Medicina Humana, 22(3). https://doi.org/10.25176/rfmh.v22i3.4753
Campo-Arias, A., Bustos-Leiton, G. J., &
Romero-Chaparro, A. (2009). Consistencia interna y dimensionalidad de la Escala
de Estrés Percibido (EEP-10 y EEP-14) en una muestra de universitarias de
Bogotá, Colombia. Aquichan, 9(3),
271–280. http://www.scielo.org.co/scielo.php?script=sci_arttext&pid=S1657-59972009000300007
Campo-Arias, A., Oviedo, H. C., & Herazo, E. (2014).
Escala de Estrés Percibido-10: Desempeño psicométrico en estudiantes de
medicina de Bucaramanga, Colombia. Revista de la Facultad de Medicina, 62(3),
407–413. https://doi.org/10.15446/revfacmed.v62n3.43735
Cao, W., Qi, X., Yao, T., Han,
X., & Feng, X. (2017). How doctors communicate the initial diagnosis of cancer matters: Cancer
disclosure and its relationship with patients’ hope and trust. Psycho-Oncology,
26(5), 640–648. https://doi.org/10.1002/pon.4063
Cardone, C., & Arnold, D. (2023). The
cancer treatment gap in lower- to middle-income countries. Oncology, 101(Suppl.
1), 2–4. https://doi.org/10.1159/000530416
Chiu, H.-Y., Huang, H.-C., Chen, P.-Y.,
Hou, W.-H., & Tsai, P.-S. (2015). Walking improves sleep in individuals
with cancer: A meta-analysis of randomized, controlled trials. Oncology
Nursing Forum, 42(2), E54–E62. https://doi.org/10.1188/15.ONF.E54-E62
Cohen, S., Kamarck,
T., & Mermelstein, R. (1983). A global measure of perceived stress. Journal
of Health and Social Behavior, 24(4), 385–396. https://doi.org/10.2307/2136404
Cook, S. A., Salmon, P., Hayes, G., Byrne,
A., & Fisher, P. L. (2018). Predictors of emotional distress a year or more
after diagnosis of cancer: A systematic review of the literature. Psycho-Oncology,
27(3), 791–801. https://doi.org/10.1002/pon.4601
Cvetkovic-Vega, A., Maguiña,
J. L., Soto, A., Lama-Valdivia, J., & López, L. E. C. (2021). Estudios transversales. Revista de la Facultad de
Medicina Humana, 21(1), 179–185. https://doi.org/10.25176/rfmh.v21i1.3069
Dalgleish, T., Black, M., Johnston, D.,
& Bevan, A. (2020). Transdiagnostic approaches to mental health problems: Current status and future directions. Journal of
Consulting and Clinical Psychology, 88(3), 179–195. https://doi.org/10.1037/ccp0000482
Duan, Y., Wang, L., Sun, Q., Liu, X.,
Ding, S., Cheng, Q., Xie, J., & Cheng, A. (2021). Prevalence and
determinants of psychological distress in adolescent and young adult patients
with cancer: A multicenter survey. Asia-Pacific Journal of Oncology Nursing,
8(3), 314–321. https://doi.org/10.4103/2347-5625.311005
Dunne, S., Mooney, O., Coffey, L., Sharp,
L., Desmond, D., Timon, C., O’Sullivan, E., & Gallagher, P. (2017).
Psychological variables associated with quality of life following primary
treatment for head and neck cancer: A systematic review of the literature from
2004 to 2015. Psycho-Oncology, 26(2), 149–160. https://doi.org/10.1002/pon.4109
EsSalud. (2020). Manual de procesos y procedimientos
de la consulta ambulatoria—Neumología [Manual]. Hospital Nacional Edgardo Rebagliati Martins. https://www.essalud.gob.pe/transparencia/procesos_procedimientos/MPP_HNERM_Consulta_Externa_Adultos_Neumologia.pdf#page=5.69
Farfán, E., & Sánchez-Villena, A. (2019). Análisis
factorial exploratorio del Inventario de Depresión de Beck (BDI-II) en
universitarios cajamarquinos. Interacciones, 5(3), e177. https://doi.org/10.24016/2019.v5n3.177
Faul, F., Erdfelder, E.,
Buchner, A., & Lang, A.-G. (2009). Statistical power analyses using G*Power 3.1: Tests
for correlation and regression analyses. Behavior Research Methods, 41(4),
1149–1160. https://doi.org/10.3758/BRM.41.4.1149
Faul, F., Erdfelder, E., Lang, A.-G.,
& Buchner, A. (2007). G*Power 3: A flexible statistical power analysis
program for the social, behavioral, and biomedical sciences. Behavior
Research Methods, 39(2), 175–191. https://doi.org/10.3758/BF03193146
Feinstein, A. R. (1970). The
pre-therapeutic classification of co-morbidity in chronic disease. Journal
of Chronic Diseases, 23(7), 455–468. https://doi.org/10.1016/0021-9681(70)90054-8
Fernández de Larrea-Baz, N., Pérez-Gómez,
B., Guerrero-Zotano, Á., Casas, A. M., Bermejo, B., Baena-Cañada, J. M.,
Antolín, S., Sánchez-Rovira, P., Ramos Vázquez, M., García-Sáenz, J. Á., Antón,
A., Muñoz, M., de Juan, A., Jara, C., Chacón, J. I., Arcusa, A., Gil-Gil, M.,
Adrover, E., Oltra, A., ... Pollán,
M. (2020). Primary breast cancer and health-related quality of life in Spanish
women: The EpiGEICAM case-control study. Scientific Reports, 10(1), 7741. https://doi.org/10.1038/s41598-020-63637-w
Fernández-de-las-Peñas, C., Rodríguez-Jiménez, J.,
Palacios-Ceña, M., de-la-Llave-Rincón, A. I.,
Fuensalida-Novo, S., Florencio, L. L., Ambite-Quesada,
S., Ortega-Santiago, R., Arias-Buría, J. L., Liew, B.
X. W., Hernández-Barrera, V., & Cigarán-Méndez,
M. (2022). Psychometric
properties of the Hospital Anxiety and Depression Scale (HADS) in previously
hospitalized COVID-19 patients. International Journal of Environmental
Research and Public Health, 19(15), 9273. https://doi.org/10.3390/ijerph19159273
Fish, J. A., Prichard, I., Ettridge, K., Grunfeld, E. A., & Wilson, C. (2015).
Psychosocial factors that influence men’s help-seeking for cancer symptoms: A
systematic synthesis of mixed methods research. Psycho-Oncology, 24(10),
1222–1232. https://doi.org/10.1002/pon.3912
Fowler, F. J. (2014). Survey research
methods. SAGE Publications.
Fuller, J. T., Hartland, M. C., Maloney,
L. T., & Davison, K. (2018). Therapeutic effects of aerobic and resistance
exercises for cancer survivors: A systematic review of meta-analyses of
clinical trials. British Journal of Sports Medicine, 52, 1–11. https://doi.org/10.1136/bjsports-2017-098285 [Verificar paginación exacta]
Galindo Vázquez, O., Benjet,
C., Juárez García, F., Rojas Castillo, E., Riveros Rosas, A., Aguilar Ponce, J.
L., Álvarez Avitia, M. Á., & Alvarado Aguilar, S. (2015). Propiedades psicométricas de la Escala Hospitalaria de
Ansiedad y Depresión (HADS) en una población de pacientes oncológicos
mexicanos. Salud Mental,
38(4), 253–258. https://doi.org/10.17711/SM.0185-3325.2015.035
Gilligan, A. M., Alberts, D. S., Roe, D.
J., & Skrepnek, G. H. (2018). Death or debt?
National estimates of financial toxicity in persons with newly diagnosed
cancer. The American Journal of Medicine, 131(10), 1187–1199.e5. https://doi.org/10.1016/j.amjmed.2018.05.020
Goebel, S., Stark, A. M., Kaup, L., von
Harscher, M., & Mehdorn, H. M. (2011). Distress in patients with newly
diagnosed brain tumours. Psycho-Oncology, 20(6),
623–630. https://doi.org/10.1002/pon.1958
González-Blanch, C., Torres, M. R., Andrés, P. C., Alfageme, O. U., Abellán, A. H., Navarro, R. M., &
Vindel, A. C. (2018). Terapia cognitivo-conductual transdiagnóstica
en atención primaria: Un contexto ideal. Revista de Psicoterapia, 29(110),
37–52.
Gordillo-León, F., Mestas-Hernández, L., Pérez-Nieto, M.
A., & Arana-Martínez, J. M. (2021). Diferencias de género en la valoración
de la intensidad emocional de las expresiones faciales de alegría y tristeza. Escritos
de Psicología, 14(1), 1–10. https://doi.org/10.24310/espsiescpsi.v14i1.12675
Grossman, M., & Wood, W. (1993). Sex differences in intensity of emotional
experience: A social role interpretation. Journal of Personality and Social
Psychology, 65(5), 1010–1022. https://doi.org/10.1037/0022-3514.65.5.1010
Gutiérrez, J. P., Jasso, V., Fuentes, A.,
Nolasco, J., Zapata, F., Castro, M., & Franco, A. (2013). Manual para la realización de encuestas de salud (Vol. 1). Instituto Nacional de Salud Pública.
Hernández-Vásquez, A., Villar Bernaola,
L., Curisinche-Rojas, M., & Timaná-Ruiz,
R. (2025). Assessment of gaps and inequalities in cancer screening at the
district level in Peru. Epidemiologia, 6(4),
74. https://doi.org/10.3390/epidemiologia6040074
Hu, Y., Vos, E. L., Baser, R. E.,
Schattner, M. A., Nishimura, M., Coit, D. G., & Strong, V. E. (2021).
Longitudinal analysis of quality-of-life recovery after gastrectomy for cancer.
Annals of Surgical Oncology, 28(1), 48–56. https://doi.org/10.1245/s10434-020-09274-z
Huo, T., Guo, Y., Shenkman, E., &
Muller, K. (2018). Assessing the reliability of the Short Form 12 (SF-12)
health survey in adults with mental health conditions: A report from the
wellness incentive and navigation (WIN) study. Health and Quality of Life
Outcomes, 16(1), 34. https://doi.org/10.1186/s12955-018-0858-2
IHME Global Burden of Disease. (2024). Share
of deaths by cause. Our World in Data. https://ourworldindata.org/grapher/share-of-deaths-by-cause
IHME Global Burden of Disease. (2024). Share
of total disease burden by cause. Our World in Data. https://ourworldindata.org/grapher/share-of-total-disease-burden-by-cause
Instituto Nacional de Enfermedades Neoplásicas. (2017). Resultados
de la implementación del programa presupuestal de control y prevención en salud
mental en el INEN, periodo 2015–2016. https://portal.inen.sld.pe/resultados-ppr-salud-mental-en-el-inen-2015-2016/
Instituto Nacional de Enfermedades Neoplásicas. (2020). Plan
de implementación y supervisión del cuidado y autocuidado de la salud mental de
los pacientes oncológicos con COVID-19 y sus familiares en el Instituto
Nacional de Enfermedades Neoplásicas [Resolución jefatural]. https://www.gob.pe/institucion/inen/normas-legales/1439674-307-2020-j-inen
Instituto Nacional de Estadística e Informática. (2014). Perfil
sociodemográfico de la zona sur de la Provincia Constitucional del Callao. https://www.inei.gob.pe/media/MenuRecursivo/publicaciones_digitales/Est/Lib1188/index.html
Instituto Nacional del Cáncer. (2015, abril 27). Estadísticas del cáncer. https://www.cancer.gov/espanol/cancer/naturaleza/estadisticas
International Agency for Research on
Cancer. (2022, January 17). Global Cancer Observatory. https://gco.iarc.fr/
Jamil, M., Merdawati,
L., Krisdianto, B. F., Khambri, D., Harahap, W. A., & Arifin, H. (2023). Determinants of
physical and mental health-related quality of life among patients with breast
cancer during COVID-19 pandemic. Nurse Media Journal of Nursing, 13(3),
263–273. https://doi.org/10.14710/nmjn.v13i3.54738
Jeffery, D. D., Ambrosio, L. A., Hopkins,
L., & Burke, H. B. (2019). Mental health comorbidities and cost/utilization
outcomes in head and neck cancer patients. Journal of Psychosocial Oncology,
37(3), 301–318. https://doi.org/10.1080/07347332.2018.1519626
Julian, L. (2011). Measures of anxiety:
State-Trait Anxiety Inventory (STAI), Beck Anxiety Inventory (BAI), and
Hospital Anxiety and Depression Scale-Anxiety (HADS-A). Arthritis Care &
Research, 63(Suppl. 11), S467–S472. https://doi.org/10.1002/acr.20561
Kim, S. J., Rha, S. Y., Song, S. K.,
Namkoong, K., Chung, H. C., Yoon, S. H., Kim, G. M., & Kim, K. R. (2011).
Prevalence and associated factors of psychological distress among Korean cancer
patients. General Hospital Psychiatry, 33(3), 246–252. https://doi.org/10.1016/j.genhosppsych.2011.02.008
Kotov, R., Krueger, R. F., Watson, D.,
Achenbach, T. M., Althoff, R. R., Bagby, R. M., Brown, T. A., Carpenter, W. T.,
Caspi, A., Clark, L. A., Eaton, N. R., Forbes, M. K., Forbush, K. T., Goldberg,
D., Hasin, D., Hyman, S. E., Ivanova, M. Y., Lynam, D. R., Markon, K., ...
Zimmerman, M. (2017). The Hierarchical Taxonomy of Psychopathology (HiTOP): A dimensional alternative to traditional nosologies. Journal of Abnormal Psychology, 126(4),
454–477. https://doi.org/10.1037/abn0000258
Kugbey, N., Oppong Asante, K., &
Meyer-Weitz, A. (2020). Depression, anxiety and quality of life among women
living with breast cancer in Ghana: Mediating roles of social support and
religiosity. Supportive Care in Cancer, 28(6), 2581–2588. https://doi.org/10.1007/s00520-019-05027-1
Lee, E.-H. (2013). Erratum to review of
the psychometric evidence of the Perceived Stress Scale [Asian Nursing
Research, 6 (2012) 121–127]. Asian Nursing Research, 7(3), 160. https://doi.org/10.1016/j.anr.2013.08.001
Ley N.° 31336, Ley Nacional del
Cáncer. (2021). http://busquedas.elperuano.pe/normaslegales/ley-nacional-del-cancer-ley-n-31336-1980284-2/
Li, Q., Lin, Y., Xu, Y., & Zhou, H.
(2018). The impact of
depression and anxiety on quality of life in Chinese cancer patient-family
caregiver dyads: A cross-sectional study. Health and Quality of Life
Outcomes, 16(1), 230. https://doi.org/10.1186/s12955-018-1051-3
Linden, W., Vodermaier,
A., Mackenzie, R., & Greig, D. (2012). Anxiety and depression after cancer
diagnosis: Prevalence rates by cancer type, gender, and age. Journal of
Affective Disorders, 141(2–3), 343–351. https://doi.org/10.1016/j.jad.2012.03.025
Lu, L., O’Sullivan, E., & Sharp, L.
(2019). Cancer-related financial hardship among head and neck cancer survivors:
Risk factors and associations with health-related quality of life. Psycho-Oncology,
28(4), 863-871. https://doi.org/10.1002/pon.5034
Ludwigson, A., Huynh, V., Bronsert, M.,
Sloan, K., Murphy, C., Christian, N., Jaiswal, K., Ahrendt, G., & Tevis, S.
E. (2020). A screening tool identifies high distress in newly diagnosed breast
cancer patients. Surgery, 168(5), 935–941. https://doi.org/10.1016/j.surg.2020.04.051
Mace, R. A., Doorley, J., Bakhshaie, J., Cohen, J. E., & Vranceanu,
A.-M. (2021). Psychological resiliency explains the relationship between
emotional distress and quality of life in neurofibromatosis. Journal of
Neuro-Oncology, 155(2), 125–132. https://doi.org/10.1007/s11060-021-03852-1
Martínez López, P., Andreu Vaillo, Y., Galdón Garrido, M. J., Romero Retes, R., García-Conde
Benet, A., & Llombart Fuertes, P. (2017). Emotional
distress and problems associated with adult oncological population. Psicooncología, 14(2–3), 217–228. https://doi.org/10.5209/PSIC.57081
Matsuda, A., Yamaoka, K., Tango, T.,
Matsuda, T., & Nishimoto, H. (2014). Effectiveness of psychoeducational
support on quality of life in early-stage breast cancer patients: A systematic
review and meta-analysis of randomized controlled trials. Quality of Life
Research, 23(1), 21–30. https://doi.org/10.1007/s11136-013-0460-3
Mejareh, Z. N., Abdollahi, B., Hoseinipalangi, Z., Jeze, M. S., Hosseinifard, H., Rafiei, S.,
Aghajani, F., Dehnad, A., Ardakani,
M. F., Ahmadi, S., Anbarhassani, H., Asl, M. T., Kan,
F. P., Aryankhesal, A., Shabaninejad,
H., Aghalou, S., & Ghashghaee, A. (2021). Global,
regional, and national prevalence of depression among cancer patients: A
systematic review and meta-analysis. Indian Journal of Psychiatry, 63(6),
527–535. https://doi.org/10.4103/indianjpsychiatry.indianjpsychiatry_77_21
Merckaert, I., Libert, Y., Delvaux, N.,
Marchal, S., Boniver, J., Etienne, A.-M., Klastersky,
J., Reynaert, C., Scalliet, P., Slachmuylder,
J.-L., & Razavi, D. (2005). Factors that influence physicians’ detection of
distress in patients with cancer: Can a communication skills training program
improve physicians’ detection? Cancer, 104(2), 411–421. https://doi.org/10.1002/cncr.21172
Ministerio de Salud. (2020). Atención de pacientes con
cáncer debe continuar durante la pandemia por COVID-19. Gob.pe. https://www.gob.pe/institucion/minsa/noticias/181037-atencion-de-pacientes-con-cancer-debe-continuar-durante-la-pandemia-por-covid-19
Ministerio de Salud. (2026). INEN casos nuevos
2000–2024 [Dashboard]. INEN. https://app.powerbi.com/view?r=eyJrIjoiMmQ2MDU0ZjMtNmIxYy00OTViLTgxZmItZmE0MGUxMmEyYWZiIiwidCI6IjU0Mjk5YmJlLWE4MzctNDVlNy1hYzljLTZjMDlmM2E2YjhkOSJ9
Mirapeix, C. (2017). La integración más allá del diagnóstico:
Aplicaciones de los nuevos modelos transdiagnósticos.
Revista de
Psicoterapia, 28(108), 15–38. https://doi.org/10.33898/rdp.v28i108.198
Nakie, G., Melkam, M., Takelle, G. M.,
Fentahun, S., Rtbey, G., Andualem, F., Kibralew, G., Wassie, Y. A., Kassa, M.
A., Tinsae, T., & Tadesse, G. (2024). Depression, anxiety and associated factors among
cancer patients in Africa: A systematic review and meta-analysis study. BMC
Psychiatry, 24, 939. https://doi.org/10.1186/s12888-024-06389-5
Ngan, T. T., Mai, V. Q., Van Minh, H.,
Donnelly, M., & O’Neill, C. (2021). Health-related quality of life among
breast cancer patients compared to cancer survivors and age-matched women in
the general population in Vietnam. Quality of Life Research, 31, 777–787.
https://doi.org/10.1007/s11136-021-02997-w
Ngo, N. T. N., Nguyen, H. T., Nguyen, P.
T. L., Vo, T. T. T., Phung, T. L., Pham, A. G., Vo, T. V., Dang, M. T. N.,
Nguyen Le Bao, T., & Duong, K. N. C. (2023). Health-related quality of life
in breast cancer patients in low- and middle-income countries in Asia: A
systematic review. Frontiers in Global Women’s Health, 4. https://doi.org/10.3389/fgwh.2023.1180383
Niedzwiedz, C. L., Knifton,
L., Robb, K. A., Katikireddi, S. V., & Smith, D.
J. (2019). Depression and anxiety among people living with and beyond cancer: A
growing clinical and research priority. BMC Cancer, 19(1), 943. https://doi.org/10.1186/s12885-019-6181-4
Nikolovski, A., Gamgoum,
L., Deol, A., Quilichini, S., Kazemir,
E., Rhodenizer, J., Brooks, D., & Alsubheen, S.
(2024). Psychometric properties of the Hospital Anxiety and Depression Scale
(HADS) in individuals with stable chronic obstructive pulmonary disease (COPD):
A systematic review. Disability and Rehabilitation, 46(7), 1230–1238. https://doi.org/10.1080/09638288.2023.2182918
Nipp, R. D., El-Jawahri,
A., Moran, S. M., D’Arpino, S. M., Johnson, P. C., Lage, D. E., Wong, R. L.,
Pirl, W. F., Traeger, L., Lennes, I. T., Cashavelly, B. J., Jackson, V. A., Greer, J. A., Ryan, D.
P., Hochberg, E. P., & Temel, J. S. (2017). The relationship between
physical and psychological symptoms and health care utilization in hospitalized
patients with advanced cancer. Cancer, 123(23), 4720–4727. https://doi.org/10.1002/cncr.30912
Organización Mundial de la Salud. (2021). Cáncer. https://www.who.int/es/news-room/fact-sheets/detail/cancer
Park, G.-R., & Kim, J. (2021).
Depressive symptoms among cancer patients: Variation by gender, cancer type,
and social engagement. Research in Nursing & Health, 44(5), 811–821.
https://doi.org/10.1002/nur.22168
Park, G.-R., & Kim, J. (2022).
Gendered trajectories of depressive symptoms and social interactions among
cancer patients. European Journal of Oncology Nursing, 56, 102092. https://doi.org/10.1016/j.ejon.2021.102092
Ploos van Amstel, F. K., van Ham, M. A. P. C.,
Peters, E. J., Prins, J. B., & Ottevanger, P. B.
(2015). Self-reported distress in patients with ovarian cancer: Is it related
to disease status? International Journal of Gynecological Cancer, 25(2),
229–235. https://doi.org/10.1097/IGC.0000000000000355
Prapa, P., Papathanasiou, I. V., Bakalis, V.,
Malli, F., Papagiannis, D., & Fradelos, E. C.
(2021). Quality of life and psychological distress of lung cancer patients
undergoing chemotherapy. World Journal of Oncology, 12(2–3), 61–66. https://doi.org/10.14740/wjon1371
Priede, A., Torres, M. R., Hoyuela, F., Herrán, A., &
González-Blanch, C. (2014). El termómetro del
distrés como prueba de cribado de ansiedad y depresión en pacientes oncológicos
recién diagnosticados. Psicooncología, 11(1), 31–43. https://doi.org/10.5209/rev_PSIC.2014.v11.n1.44915
Ravindran, O., Shankar, A., & Murthy, T. (2019). A comparative study on perceived stress,
coping, quality of life, and hopelessness between cancer patients and
survivors. Indian Journal of Palliative Care, 25(3), 414–420. https://doi.org/10.4103/IJPC.IJPC_1_19
Riba, M. B., Donovan, K. A., Andersen, B.,
Braun, I., Breitbart, W. S., Brewer, B. W., Buchmann, L. O., Clark, M. M.,
Collins, M., Corbett, C., Fleishman, S., Garcia, S., Greenberg, D. B., Handzo,
R. G. F., Hoofring, L., Huang, C., Lally, R., Martin,
S., McGuffey, L., Mitchell, W., Morrison, L. J., Pailler,
M., Palesh, O., Parnes, F., Pazar, J. P., Ralston,
L., Salman, J., Shannon-Dudley, M. M., Valentine, A. D., McMillian, N. R.,
& Darlow, S. D. (2019). Distress management, version 3.2019, NCCN Clinical
Practice Guidelines in Oncology. Journal of the National Comprehensive
Cancer Network, 17(10), 1229–1249. https://doi.org/10.6004/jnccn.2019.0048
Sarfati, D., Koczwara, B., & Jackson,
C. (2016). The impact of comorbidity on cancer and its treatment. CA: A
Cancer Journal for Clinicians, 66(4), 337–350. https://doi.org/10.3322/caac.21342
Schmidt, S., Vilagut,
G., Garin, O., Cunillera, O., Tresserras, R., Brugulat, P., Mompart, A.,
Medina, A., Ferrer, M., & Alonso, J. (2012). Normas de referencia para el Cuestionario de Salud SF-12 versión 2 basadas
en población general de Cataluña. Medicina Clínica, 139(14), 613–625. https://doi.org/10.1016/j.medcli.2011.10.024
Seedhom, A. E., Kamel, E. G., Mohammed, E. S.,
& Raouf, N. R. (2019). Predictors of perceived stress among medical and
nonmedical college students, Minia, Egypt. International Journal of
Preventive Medicine, 10, 107. https://doi.org/10.4103/ijpvm.IJPVM_6_18
Smarr, K., & Keefer, A. (2011).
Measures of depression and depressive symptoms: Beck Depression Inventory-II
(BDI-II), Center for Epidemiologic Studies Depression Scale (CES-D), Geriatric
Depression Scale (GDS), Hospital Anxiety and Depression Scale (HADS), and
Patient Health Questionnaire-9 (PHQ-9). Arthritis Care & Research, 63(Suppl.
11), S454–S466. https://doi.org/10.1002/acr.20556
Suresh, K., & Chandrashekara, S.
(2012). Sample size estimation and power analysis for clinical research
studies. Journal of Human Reproductive Sciences, 5(1), 7–13. https://doi.org/10.4103/0974-1208.97779
Tejero, A. (1986). Uso clínico del HAD (Hospital Anxiety
and Depression Scale) en población psiquiátrica: Un
estudio de su sensibilidad, fiabilidad y validez. Revista
del Departamento de Psiquiatría de la Facultad de Medicina de Barcelona, 13, 233-238
Terol-Cantero, M. C., Cabrera-Perona, V., &
Martín-Aragón, M. (2015). Revisión de estudios de la Escala de Ansiedad y
Depresión Hospitalaria (HAD) en muestras españolas. Anales de Psicología, 31(2),
494–503. https://doi.org/10.6018/analesps.31.2.172701
Tortella-Feliu, M. R. (2014). Los trastornos de ansiedad en el
DSM-5. Cuadernos de Medicina Psicosomática y Psiquiatría de Enlace, 110,
62–69.
Trayes, K. P., & Cokenakes,
S. E. H. (2021). Breast cancer
treatment. American Family Physician, 104(2), 171–178.
Villarreal-Zegarra, D., Cabrera-Alva, M.,
Carrillo-Larco, R. M., & Bernabe-Ortiz, A. (2020). Trends in the prevalence
and treatment of depressive symptoms in Peru: A population-based study. BMJ Open, 10(7), e036777. https://doi.org/10.1136/bmjopen-2020-036777
Vizioli, N. A., & Pagano, A. E.
(2020). Adaptación del Inventario
de Ansiedad de Beck en población de Buenos Aires. Interacciones, 6(3), e171. https://doi.org/10.24016/2020.v6n3.171
Vodermaier, A., & Millman, R. D. (2011). Accuracy
of the Hospital Anxiety and Depression Scale as a screening tool in cancer
patients: A systematic review and meta-analysis. Supportive Care in Cancer,
19(12), 1899–1908. https://doi.org/10.1007/s00520-011-1251-4
Vy, H. T., Nghia, D. Q., Nguyen, L. T. N.,
Binh, V. T. T., Minh, H. T. T., Kinh, H. D., Nam, T. L. T. T., Bang, H. T., & Trung, L. Q. (2025).
Psychosocial support needs and associated factors among cancer patients: A
hospital-based, single-center cross-sectional study in Vietnam. Asian
Pacific Journal of Cancer Prevention, 26(7), 2619–2626. https://doi.org/10.31557/APJCP.2025.26.7.2619
Walker, Z. J., Xue, S., Jones, M. P.,
& Ravindran, A. V. (2021). Depression, anxiety, and other mental disorders
in patients with cancer in low- and lower-middle-income countries: A systematic
review and meta-analysis. JCO Global Oncology, 7, 1233–1250. https://doi.org/10.1200/GO.21.00056
Wang, S.-B., Qin, S.-H., Li, X.-M., Li,
W.-L., & Lu, T.-Q. (2018). Study of psychological distress and quality of
life in patients with oral cancer. Shanghai Journal of Stomatology, 27(5),
486–490. https://10.19439/j.sjos.2018.05.008
Weber, D., & O’Brien, K. (2017).
Cancer and cancer-related fatigue and the interrelationships with depression,
stress, and inflammation. Journal of Evidence-Based Complementary &
Alternative Medicine, 22(3), 502–512. https://doi.org/10.1177/2156587216676122
Wen, S., Xiao, H., & Yang, Y. (2019).
The risk factors for depression in cancer patients undergoing chemotherapy: A
systematic review. Supportive Care in Cancer, 27(1), 57–67. https://doi.org/10.1007/s00520-018-4466-9
Yeh, Y.-C., Sun, J.-L., & Lu, C.-H.
(2021). Associations between perceived stress and quality of life in gynaecologic cancer patient-family caregiver dyads. European
Journal of Oncology Nursing, 55, 102060. https://doi.org/10.1016/j.ejon.2021.102060
Zhuang, H., Ma, Y., Wang, L., & Zhang,
H. (2018). Effect of early palliative care on quality of life in patients with
non-small-cell lung cancer. Current Oncology, 25(1), e54–e58. https://doi.org/10.3747/co.25.3639
Zigmond, A. S., & Snaith, R. P.
(1983). The Hospital Anxiety and Depression Scale. Acta Psychiatrica Scandinavica, 67(6), 361–370. https://doi.org/10.1111/j.1600-0447.1983.tb09716.x